Fortimel Forticare Sensations is a food for special medical purposes for the dietary management of patients with or at risk of disease related malnutrition due to cancer, chronic catabolic disease or cachexia.
Fortimel Compact Protein Sensations is a food for special medical purposes for the dietary management of disease related malnutrition. Must be used under medical supervision. Our SENSATIONS range is made of specially designed flavours for patients with taste alterations. They provide a cool or warm taste sensation in the mouth.
World Cancer Day: Closing the Nutritional Care Gap!
Nutritional Intervention for Quality of Life and Treatment Outcomes. Why it matters?
Nutritional intervention can improve both quality of life and treatment outcomes for cancer patients1. Most patients with cancer experience metabolic and psychological stress, sensory alterations, poor appetite and malabsorption, resulting in malnutrition2-4.
Up to 70% of patients with cancer experience taste and smell changes2. Sensory alterations can lead to a reduction in food intake and result in increased weight loss2,3. Learn more about taste alterations:
Weight and muscle loss due to malnutrition in patients with cancer negatively impacts clinical outcomes such as post-surgery recovery, treatment tolerance and quality of life5. Nutritional support in patients with cancer can ensure optimal treatment outcomes and improve quality of life1.
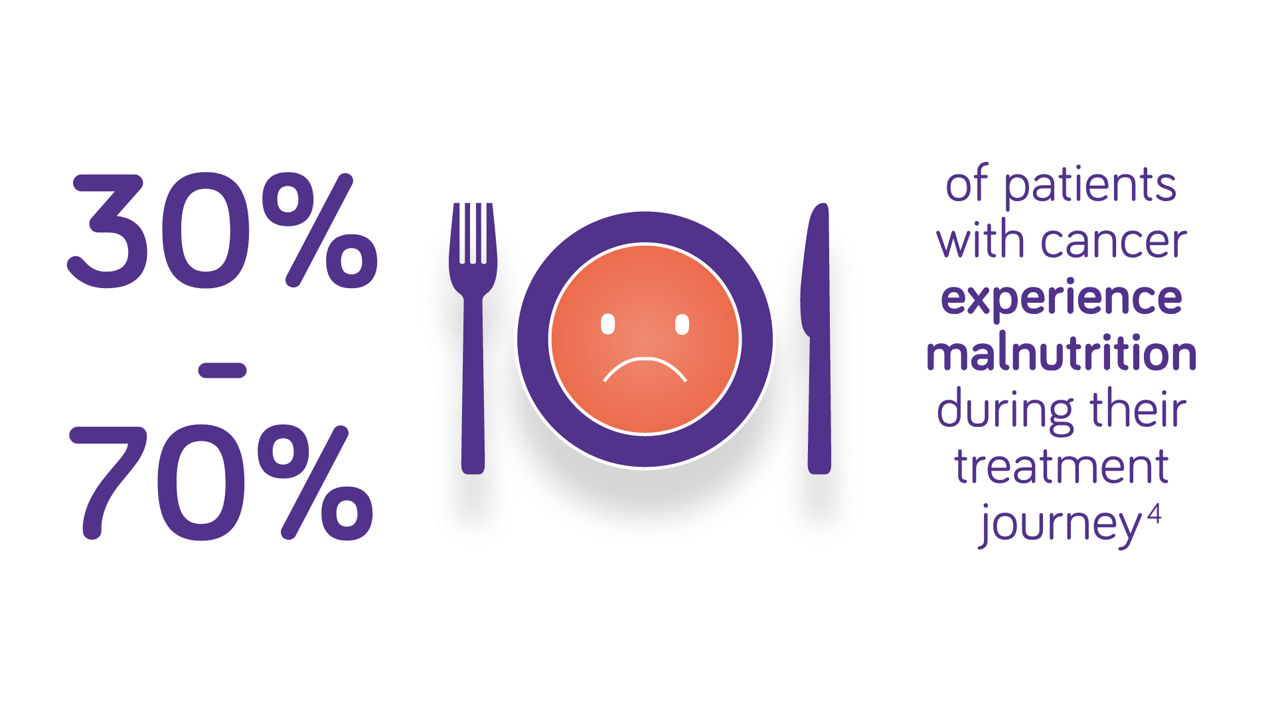
Guidelines recommend early assessment of nutritional risk in patients with cancer
ESPEN and ESMO guidelines recommend routine screening from diagnosis onwards to detect nutritional imbalances in patients with cancer6,7.
According to ESMO and ESPEN, Oral Nutritional Supplements (ONS) can be supplied to improve energy intake and induce weight gain when nutritional needs can’t be met via food alone.
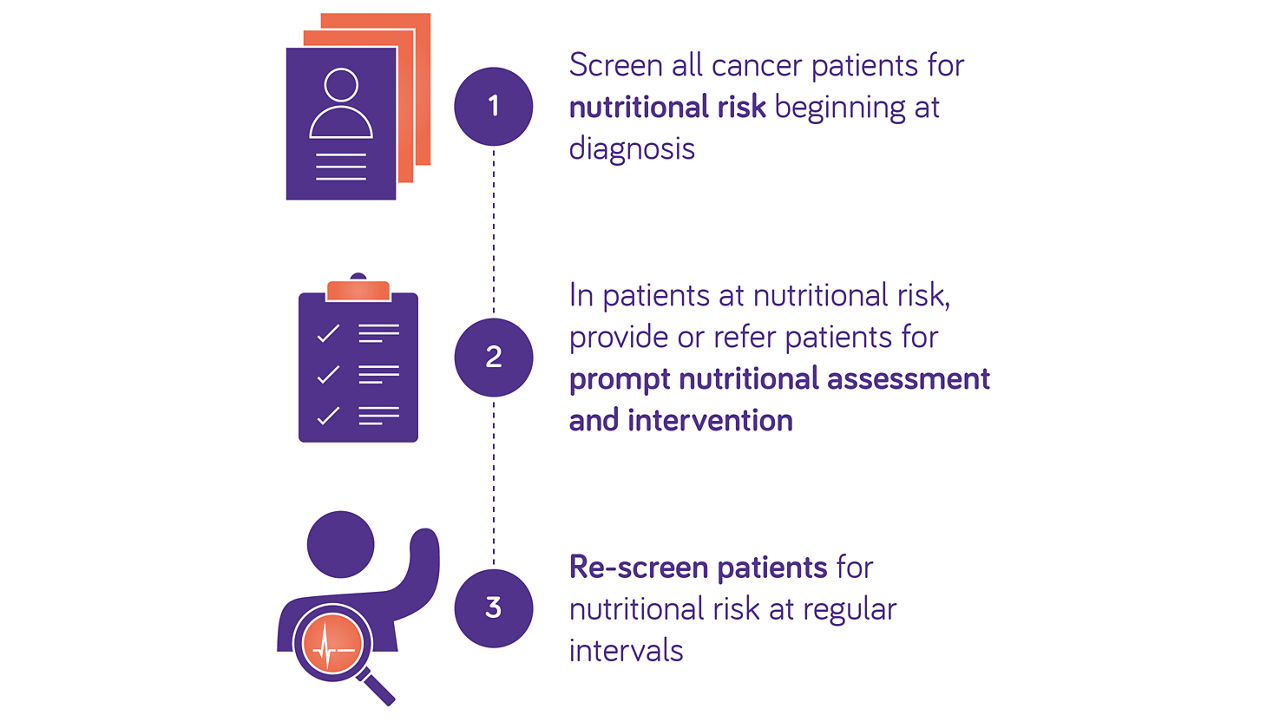
Nutritional intake, weight changes, body mass index, muscle mass and systemic inflammation should be evaluated6,7.
Routine screening for malnutrition allows early nutritional intervention and prehabilitation strategies to prevent nutritional decline, in patients with cancer.
Read more on ESMO guidelines for management of cancer cachexia:
The impact of weight loss in treatment outcomes

Cancer-related weight loss is associated with poor clinical outcomes and lower quality of life. Did you know that 3 out 5 oncology patients report weight loss during their cancer journey and almost 1 out of 3 patients claim that weight loss negatively impacted their treatment plan including delays, modifications and interruptions?13
Learn more about weight loss and nutrition-related challenges from oncology patient perspective.
Learn more about the consequences of weight loss during the cancer journey:
Cancer-related malnutrition and/or low muscle mass (sarcopenia) may lead to suboptimal anti-cancer treatment outcomes8,9. Loss of muscle mass is a major component of weight loss in cancer-related malnutrition and cachexia and may represent > 70% of total body mass lost by a cancer patient11. Lower muscle mass is a significant, independent predictor of early treatment termination and dose reductions10, and in patients receiving cancer immunotherapy, the risk of high-grade adverse events is increased >5 fold in patients with low muscle mass and/or low muscle attenuation*,12.
ESMO recommends early nutritional support including medical nutrition as an integral part of cancer care14.
Discover our tailored portfolio addressing the evolving nutritional needs of patients with cancer
Nutricia products are food for special medical purposes and must be used under medical supervision.

Join us in closing the nutritional care gap for cancer patients, empowering them with knowledge, support, and tailored nutritional care throughout their journey.
Take a look at the World Cancer Day Organization (WCD website) to learn more and discover how to take action.
Together, we can enhance the well-being of those on the cancer journey.
- Laviano A, et al. Proc Nutr Soc. 2018;77(4):388-93.
- Weimann A, et al. Clin Nutr. 2017;36(3):623-50.
- Spotten LE, et al. Ann Oncol. 2017;28(5):969-84.
- Brisbois TD, et al. J Pain Symptom Manage. 2011;41(4):673-83.
- Ryan AM, et al. Nutrition. 2019;67-68:110539.
- Arends J, et al. Clin Nutr. 2017;36(1):11-48.
- Arends J, et al. ESMO Open. 2021;6(3):100092.
- Anjanappa M, et al. Tech Innov Patient Support Radiat Oncol. 2020;16:50-7.
- Cruz-Jentoft AJ, et al. Age Ageing. 2019;48(1):16-31.
- Cespedes Feliciano EM, et al. Cancer 2017;23:4868–77.
- Silver HJ, et al. Head & Neck. 2007;29(10):893-900.
- Daly LE, et al. Br J Cancer. 2017;116(3):310-7.
- Ipsos European Oncology Patient Survey, 2023. Data on file
- Arends J, et al. 2021, ESMO Open; 6(3):100092
Are you a healthcare professional or (carer of) a diagnosed patient?
The product information for this area of specialization is intended for healthcare professionals or (carers of) diagnosed patients only, as these products are for use under healthcare professional supervision.
Please click ‘Yes’ if you are a healthcare professional or (carer of) a diagnosed patient, or ‘No’ to be taken to a full list of our products.
The information on this page is intended for healthcare professionals only.
If you aren't a healthcare professional, you can visit the page with general information, by clicking 'I'm not a healthcare professional' below.



